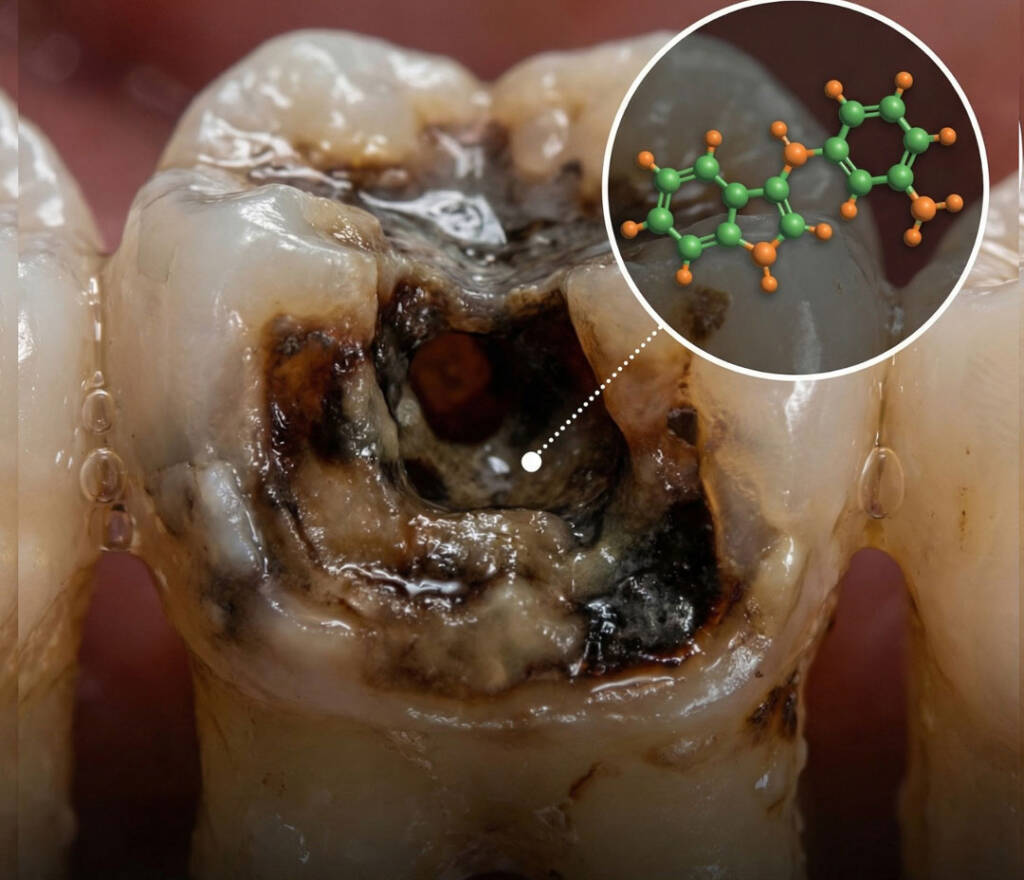
Cutting-edge research suggests a molecule from cruciferous vegetables may tackle the root cause of tooth decay — bacterial biofilms — with precision and low toxicity.
Dental caries (tooth decay) remains a persistent global health issue. Traditional oral care focuses on mechanical removal of plaque and the use of broad-spectrum antiseptics. But new research shows a natural compound, 3,3′-Diindolylmethane (DIM), may directly disrupt the mechanisms by which cavity-causing bacteria protect themselves, opening the door to smarter and gentler approaches to dental health.
What Is DIM?
3,3′-Diindolylmethane (DIM) is a bioactive compound produced when the body breaks down indole-3-carbinol, which is abundant in cruciferous vegetables such as broccoli, cabbage, kale, and Brussels sprouts.
DIM has been studied in other fields of health science — notably cancer research — where it exhibits anti-proliferative and anti-inflammatory effects. Its impact on microbial behavior, particularly biofilms, is an exciting new frontier.
DIM vs. Streptococcus mutans: A New Anti-Biofilm Strategy
The bacterium Streptococcus mutans is a key driver of dental caries. It adheres to tooth surfaces and produces biofilms — complex, sticky 3D structures that form the basis of dental plaque. These biofilms are resilient and difficult to remove with brushing alone and contribute substantially to enamel demineralization through acid production when carbohydrates are metabolized.
How DIM Works Against Biofilms
A 2023 study published in Antibiotics found that DIM significantly interferes with the formation of S. mutans biofilms in laboratory settings:
- Acid resilience reduction: The durability of biofilms under acidic conditions — a major factor in enamel erosion — was significantly decreased.
- Biofilm reduction: At an optimal concentration (0.5 µM), DIM reduced biofilm formation by up to 92% compared to untreated controls.
- EPS suppression: DIM lowered production of extracellular polymeric substances (EPS), the sticky glue-like material that anchors bacteria and stabilizes biofilms.
Crucially, DIM did not kill the bacteria directly at these concentrations; instead, it prevented them from forming their protective shield. This anti-virulence mechanism means DIM could reduce pathogenic behavior without exerting strong selective pressure that leads to resistance.
Lab Conditions vs. Real-World Oral Cavities
The study also evaluated DIM’s activity under conditions that mimic the hydrodynamics of the oral environment (with fluid flow similar to saliva):
- DIM treatment reduced S. mutans biofilm volume by about 50% in these dynamic systems.
- There was no significant increase in dead bacteria, reinforcing that DIM’s main effect is on biofilm structure, not bacterial viability.
While promising, these results remain in vitro (laboratory) — meaning DIM’s performance in the human mouth or products like toothpaste has yet to be tested in clinical trials.
Why DIM’s Mechanism Matters
Current antimicrobial agents in toothpaste and mouthwash (chlorhexidine, essential oils, etc.) often act broadly, disrupting beneficial microbes along with harmful ones. This can negatively affect the oral microbiome and contribute to side effects like staining or altered taste.
DIM’s targeted anti-biofilm action, focusing on virulence factors rather than bacterial death, represents a precision approach:
✔️ Preserves beneficial oral flora
✔️ Reduces plaque formation at the source
✔️ Less likely to promote resistance
This strategy aligns with modern microbiome-friendly therapeutic development across medicine.
Broader Antimicrobial Biofilm Activity
Beyond S. mutans, other studies show DIM can disrupt biofilm formation by a range of bacteria:
- It inhibited acne-associated bacterial and fungal biofilms involving Cutibacterium acnes, Staphylococcus aureus, and Candida albicans.
- DIM also reduced virulence and biofilm formation in major pathogens Pseudomonas aeruginosa and Acinetobacter baumannii when tested in other research contexts.
These findings suggest DIM’s antibiofilm effect is not limited to oral bacteria, supporting its potential as a broad-spectrum anti-biofilm agent.
Current Limitations and Next Steps
While DIM’s anti-biofilm potential is clear in lab studies, there are important caveats:
🧪 No human clinical trials yet — DIM’s impact on actual tooth decay, plaque prevention, and oral microbiome balance in people remains untested.
🧪 Effectiveness depends on formulation and concentration — the precise dose that works in the mouth may differ from laboratory conditions.
🧪 Food consumption of cruciferous vegetables alone likely insufficient — therapeutic concentrations rely on isolated DIM or enriched formulations, not diet by itself.
Researchers propose future work should focus on:
- In-vivo animal studies
- Human clinical trials
- Safety and stability testing in dental product formulations
- Synergistic effects with fluoride and other cariostatic agents
Conclusion: A Promising Natural Ally in Dental Care
DIM — a naturally derived compound from cruciferous vegetables — shows strong potential as a anti-biofilm agent against the primary cavity-causing bacterium, S. mutans. Its mechanism of inhibiting biofilm formation without killing bacteria outright aligns with modern, microbiome-friendly therapeutic approaches.
While still early in the research pipeline, DIM could become a key ingredient in future oral-care products that target plaque formation at the molecular level, offering a more effective and gentle means of cavity prevention.
Scientific References
- Yifat Baruch et al. 3,3′-Diindolylmethane (DIM): A Potential Therapeutic Agent against Cariogenic Streptococcus mutans Biofilm. Antibiotics (2023). MDPI
- PubMed summary of DIM biofilm study. PubMed
- Antibiofilm and antimicrobial activity of DIM against acne-associated organisms. PubMed
- Anti-virulence biofilm study on major bacterial pathogens. PubMed
- SciTechDaily overview and research context (experimental phase). SciTechDaily